UDI Requirements for Engineering Polymers
Critical UDI Specifications for Medical Polymers
Medical device UDI marking on polymers must meet specific requirements that are directly impacted by laser-material physics:
Minimum Module Size Constraints
Durability Requirements
- 25+ autoclave cycles at 134°C
- Gamma sterilization resistance
- Chemical cleaning compatibility
- No subsurface degradation
- Consistent contrast maintenance
UDI Failure Modes in Polymers
Immediate Failures
- Insufficient contrast due to foaming
- Module distortion from heat zones
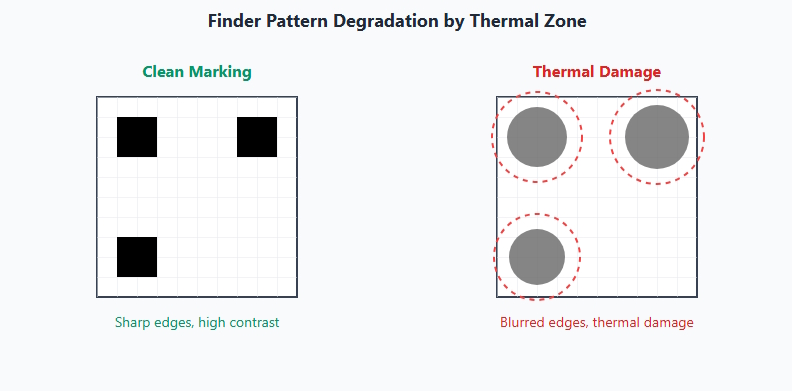
- Finder pattern geometry errors
- Edge definition loss
Long-term Failures
- Mark fading after sterilization
- Subsurface crack propagation
- Contrast degradation over time
- Environmental stress cracking